O-Demethylation
![]()
Common Conditions:
BBr3
Tribromoborane (BBr3) is a relatively mild reagent for effectively cleaving phenolic methyl ethers. The reaction is typically performed in DCM with cooling or at RT.
![]()
HBr
Strong protic acids such as hydrobromic acid (HBr) can cleave phenolic methyl ethers. The reaction is usually done at elevated temperatures resulting in relatively low functional group tolerance relative to milder methods such as BBr3.
![]()
Thiolates
Strong nucleophiles such as thiolates can nucleophilicly demethylate phenolic methyl ethers. Reactions are typically done in a polar aprotic solvent (ex. DMF) at elevated temperatures.
![]()
Reaction Map:
The reaction map is intended to provide insight into possible reactions one step before and after the title reaction. It also serves as an alternative way to navigate the website, and as a means of coming up with retrosynthetic ideas. Click on the reaction arrow to visit the page.
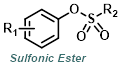 |
||
 |
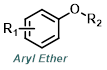 |
|
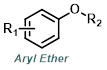 |
||
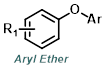 |
References:
1) Kocienski, P. J.; Protecting Groups, 3rd Edition
