Boc Protection
(Boc2O + Base)
Examples:
Example 1

To a solution of the SM (1 g, 2.23 mmol) and TEA (1.24 mL, 8.93 mmol) in DCM (20 mL) under N2 was added (Boc)2O (1.46 g, 6.7 mmol) at 5-10 C. The reaction mixture was stirred overnight at RT, after which time it was diluted with H2O (20 mL) and extracted with DCM (2 x 20 mL). The combined org extracts were washed with H2O (2 x 20 mL), brine, dried (Na2SO4), and concentrated to provide the product as a white solid. [1.2 g, 83%]
[Patent Reference: WO2010038081, page 112, ![]() (33.8 MB)]
(33.8 MB)]
Example 2

To a solution of the SM (15 g, 43 mmol) and (Boc)2O (23 g, 105 mmol) in DCM (150 mL) was added TEA (19 mL, 140 mmol). The reaction mixture was stirred at RT for 4 h, after which time the solvents were removed in vacuo and the residue was purified by silica gel chromatography (5-10% EtOAc in petroleum ether) to provide the product as a colorless solid. This material was used directly in the following step. [25 g]
[Patent Reference: WO2014177977, page 65, ![]() (6.0 MB)]
(6.0 MB)]
Example 3
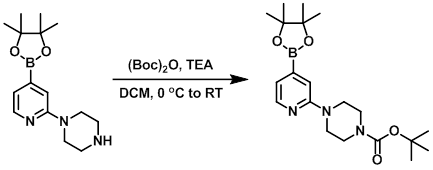
A solution of the SM (0.5 g, 1.73 mmol) in DCM (13 mL) was cooled to 0 C and treated with TEA (0.265 mL, 1.90 mmol) followed by (Boc)2O (377 mg, 1.73 mmol). The reaction mixture was slowly warmed to RT. Upon completion, the reaction mixture was washed with H2O, dried (MgSO4), and concentrated. The resulting material was dried under vacuum at RT overnight to provide the product as a white solid. [606 mg, 90%]
[Patent Reference: WO2010027500, page 105, ![]() (22.8 MB)]
(22.8 MB)]
Example 4

To a slurry of the SM (106.1 g, 517 mmol) and DIEA (108 mL, 621 mmol) in DCM (1 L) was added a solution of (Boc)2O (119 g, 543 mmol) in DCM (100 mL) via an addition funnel over 1 h. The reaction mixture was stirred at RT for 1 h, after which time the reaction mixture was concentrated in vacuo. The resulting crude material was dissolved in EtOAc (1 L) and washed with H2O (3 x 300 mL), then brine (300 mL), and dried (MgSO4). The solvent was removed in vacuo to provide the product as an off-white solid. [139 g, 100%]
[Patent Reference: WO2012129344, page 126, ![]() (7.3 MB)]
(7.3 MB)]
Example 5
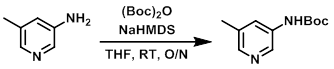
To a solution of the SM (1.0 equiv) in THF (0.5M) at RT was added 1M NaHMDS in THF (2.2 equiv). The mixture was stirred at RT for 15 min then treated with (Boc)2O (1.05 equiv) in THF. The resulting reaction mixture was stirred at RT overnight, after which time it was concentrated. The concentrate was partitioned between 0.2M HCl (60 mL) and EtOAc. The org layer was separated and washed with saturated aq NaHCO3, brine, dried (Na2SO4), concentrated, and purified by silica gel flash chromatography (40% EtOAc/hexane) to provide the product as a yellow solid. [88%]
[Patent Reference: WO2010026121, page 66, ![]() (3.6 MB)]
(3.6 MB)]
Example 6
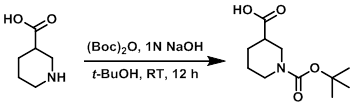
To a solution of the SM (50.0 g, 387.5 mmol) in t-BuOH (464 mL) at 0 C was added 1N NaOH (464 mL). After stirring for 10 min, the mixture was treated with (Boc)2O (143.6 g, 659.0 mmol). The reaction mixture was stirred at RT for 12 h, after which time it was concentrated in vacuo, and neutralized with 1N HCl. The resulting solids were filtered and dried to provide the product as a white solid. [86.0 g, 97%]
[Patent Reference: WO2014149164, page 213, ![]() (23.7 MB)]
(23.7 MB)]
Example 7
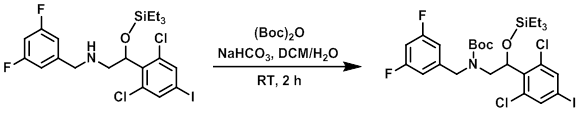
To a solution of the SM (1.87 g, 3.26 mmol) in 4:1 DCM/H2O (20 mL) was added NaHCO3 (0.55 g, 6.5 mmol) and (Boc)2O (1.07 g, 4.9 mmol) in DCM (8 mL) at 0 C. The reaction mixture was stirred at RT for 2 h, after which time it was quenched with H2O (100 mL) and extracted with DCM (2 x 30 mL). The combined organics were washed with H2O (50 mL), brine (50 mL), dried (Na2SO4), and concentrated to provide the product as a colorless gum. [2.47 g, crude]
[Patent Reference: WO2015129926, page 76, ![]() (21.5 MB)]
(21.5 MB)]
Example 8
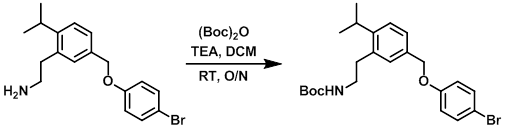
(Boc)2O (203 mg, 0.93 mmol) and TEA (0.35 mL, 2.5 mmol) were added to a suspension of the SM (295 mg, 0.85 mmol) in DCM (4.5 mL). The reaction mixture was stirred at RT overnight. The mixture was diluted with DCM and quenched with H2O. The org layer was separated, washed with H2O, NaHCO3, dried (MgSO4), and concentrated to provide the product as a colorless oil. [300 mg, 79%]
[Patent Reference: WO2015144799, page 179, ![]() (18.8 MB)]
(18.8 MB)]
