TEA
(Triethylamine)
General Information:
Structure:
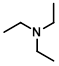
CAS Number: 121-44-8
Molecular Weight: 101.19 g/mol
Appearance: Colorless liquid
Triethylamine (TEA) is a very commonly used organic base. Diisopropylethylamine (DIEA) is a closely related organic base. DIEA is more sterically hindered than TEA therefore is less prone to quaternization when used with highly reactive alkylation agents. TEA has a boiling point of 89 C, making it easier to remove via rotovap distillation. DIEA has a boiling point of 127 C, making it more useful for reactions that are over 90 C. In most situations TEA and DIEA can be used interchangably. However, for certain situations one is a better choice than the other.
Common Uses:
- Base in Swern oxidations
- Base in Parikh-Doering oxidations
- Base in Corey-Kim oxidations
- Base in Pd catalyzed reactions (Sonogashira or Heck)
- Base in substitution reactions
- Base in SNAr reactions
Safety:
Triethylamine is a corrosive, flammable liquid.
References:
1) Wikipedia: Triethylamine (link)
2) www.sigmaaldrich.com: Triethylamine (link)
3) Reich, H. J.; Rigby, J. H.; Handbook of Reagents for Organic Synthesis, Acidic and Basic Reagents
