Amine to Amide
(EDC + HOBt)
Examples:
Example 1

To a solution of the SM (1.0 g, 6.4 mmol) in DMF (5.0 mL) at 0 C was added EDC-HCl (1.2 g, 7.7 mmol), NH4Cl (1.4 g, 26.9 mmol), and HOBt (1.1 g, 8.3 mmol). The reaction mixture was stirred at RT for 12 h, after which time ice-cold H2O was added and the mixture was extracted with EtOAc. The org layer was dried (Na2SO4) and concentrated in vacuo to provide the product as an orange/red solid. [600 mg, 61%]
[Patent Reference: WO2014149164, page 227, ![]() (23.7 MB)]
(23.7 MB)]
Example 2
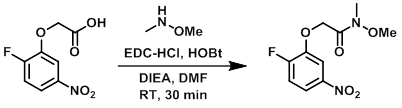
To a solution of the acid (700 mg, 3.2 mmol) in DMF (10 mL) was added DIEA (1.25 g, 9.7 mmol). The mixture was cooled to 0 C and treated with EDC-HCl (1.2 g, 6.5 mmol), HOBt (879 mg, 6.5 mmol), and the amine (473 mg, 4.9 mmol). The reaction was stirred at RT for 30 min. After completion, the mixture was diluted with H2O (50 mL) and extracted with EtOAc (50 mL). The org layer was dried (Na2SO4) and concentrated in vacuo. The resulting material was purified by silica gel column chromatography to provide the product as a gummy liquid. [400 mg, 83%]
[Patent Reference: WO2014149164, page 337, ![]() (23.7 MB)]
(23.7 MB)]
Example 3
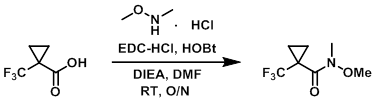
To a mixture of the acid (150 mg, 0.974 mmol), HOBt (224 mg, 1.46 mmol), EDC-HCl (280 mg, 1.46 mmol), and the amine (142 mg, 1.46 mmol) in DMF (5 mL) was added DIEA (0.50 mL, 2.92 mmol). The reaction mixture was stirred at RT overnight. The mixture was quenched with H2O (30 mL) and extracted with EtOAc. The org layer was washed with H2O, brine, dried (MgSO4), and concentrated in vacuo to provide the product as a pale yellow oil. [164 mg, 85%]
[Patent Reference: WO2015129926, page 93, ![]() (21.5 MB)]
(21.5 MB)]
Example 4

EDCI (1.2 g, 6.2 mmol) was added to a solution of the SM (0.8 g, 4.1 mmol), HOBt (0.8 g, 6.2 mmol), DIEA (1 mL, 6.2 mmol), and methylamine (0.46 mL, 5.3 mmol) in DCM (25 mL). The reaction mixture was stirred at RT for 8 h. H2O and DCM were added. The layers were separated and the org layer was dried (MgSO4) and concentrated. The resulting material was purified by Prep LC (24 g silica, 2% MeOH/DCM) to provide the product. [500 mg, 59%]
[Patent Reference: WO2015144799, page 321, ![]() (18.8 MB)]
(18.8 MB)]
Example 5
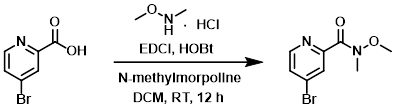
To a mixture of the acid (1.01 g, 5.0 mmol), the amine (536 mg, 5.5 mmol), and N-methylmorpholine (2.02 g, 20 mmol), in DCM (20 mL) was added EDCI (959 mg, 5.0 mmol) and HOBt (676 mg, 5.0 mmol) in portions at 0 C. The reaction mixture was stirred at RT for 12 h. The mixture was extracted with DCM (3 x 30 mL). The combined organics were washed with brine, dried (Na2SO4), and concentrated. The resulting crude material was purified by silica gel chromatography (10:1 PE/EtOAc) to provide the product as a colorless oil. [545 mg, 44%]
[Patent Reference: WO2016011390, page 148, ![]() (20.2 MB)]
(20.2 MB)]
