Aldehyde to Alcohol
(NaBH4)
Examples:
Example 1
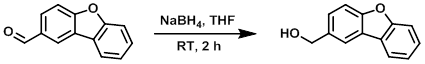
To a solution of the SM (191 mg, 0.973 mmol) in THF (10 mL) was added NaBH4 (37 mg, 0.978 mmol). The mixture was stirred at RT for 2 h, after which time it was quenched with aq 1N HCl (10 mL) and stirred for 20 min. The resulting mixture was then extracted with DCM (2 x 50 mL). The organic phase was washed with H2O (10 mL), brine (10 mL), dried (Na2SO4), and concentrated in vacuo to provide to product as an off-white solid. [192 mg, 100%]
[Patent Reference: WO2012112946, page 50, ![]() (11.2 MB)]
(11.2 MB)]
Example 2
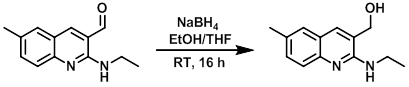
To a solution of the SM (500 mg, 2.33 mmol) in EtOH/THF (7:3, 35 mL) at 0 C was added NaBH4 (88 mg, 2.33 mmol) in small portions. The reaction mixture was stirred at RT overnight, after which time it was cooled to 0 C and treated with 6N HCl (6 mL). The resulting mixture was then neutralized with 2N NaOH until pH 7 and concentrated to dryness. The crude residue was diluted with DCM (50 mL), washed with brine (3 x 50 mL), dried (Na2SO4), and concentrated to provide the product as a yellow solid. [460 mg, 91%]
[Patent Reference: WO2012112946, page 111, ![]() (11.2 MB)]
(11.2 MB)]
Example 3
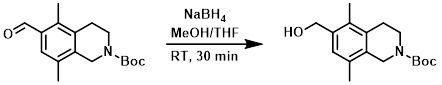
The SM (785 mg, 2.71 mmol) was dissolved in THF (5.0 mL) and MeOH (5.0 mL). The mixture was cooled to 0 C and treated with NaBH4 (156 mg, 4.07 mmol). The reaction mixture was stirred at RT for 30 min. Excess reagents were consumed by the addition of aq NH4Cl and the mixture was stirred at RT for 10 min. The mixture was extracted with EtOAc. The org layer was washed with aq NH4Cl, brine, dried (MgSO4), and concentrated. The resulting material was purified by silica gel flash chromatography to provide the product as a white solid. [626 mg, 79%]
[Patent Reference: WO2016014463, page 99, ![]() (6.7 MB)]
(6.7 MB)]
Example 4
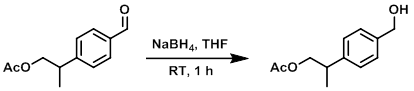
A solution of the SM (3.70 g, 17.9 mmol) in THF (40 mL) was treated with NaBH4 (1.36 g, 35.9 mmol) and stirred at RT for 1 h. The mixture was diluted with DCM and H2O. The org layer was separated, washed with brine, dried (MgSO4), and concentrated to give 3.6 g of material. The material was purified by Prep LC (120 g silica, 40-80% EtOAc/heptane) to provide the product as a yellow oil. [2.19 g, 59%]
[Patent Reference: WO2015144799, page 229, ![]() (18.8 MB)]
(18.8 MB)]
