Reductive Amination
(NaCNBH3)
Examples:
Example 1
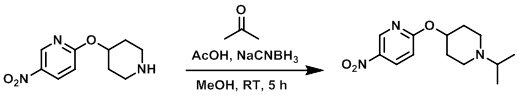
To a solution of 10% AcOH in MeOH was added the SM (1 equiv) and dry acetone (5 equiv). The solution was stirred at RT 1 h, after which time it was cooled to 0 C and treated with NaCNBH3 (1.5 equiv). The reaction was stirred at RT for 5 h. The mixture was concentrated and the residue brought to pH = 10 using Na2CO3. The mixture was extracted with EtOAc and the org layer was washed with H2O, brine, dried (Na2SO4), and concentrated to dryness. The crude material was purified by silica gel column chromatography (2% MeOH/DCM) to provide the pdt as a yellow solid.
[Patent Reference: WO2007084786, page 112, ![]() (9.4 MB)]
(9.4 MB)]
Example 2
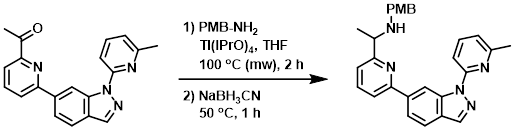
A mixture of the SM (260 mg, 0.79 mmol), PMB-NH2 (162 mg, 1.18 mmol), and Ti(iPrO)4 (168 mg, 1.58 mmol) in THF (10 mL) was stirred in a microwave reactor at 100 C for 2 h. After cooling to RT, NaBH3CN (98 mg, 1.58 mmol) was added to the mixture. The reaction mixture was stirred at 50 C for 1 h. The mixture was poured into H2O (10 mL) and extracted with EtOAc (20 mL). The org phase was concentrated and the residue was purified by silica gel column chromatography (6:1 PE/EtOAc) to provide the product as a yellow solid. [180 mg, 50%]
[Patent Reference: WO2016011390, page 104, ![]() (20.2 MB)]
(20.2 MB)]
Example 3
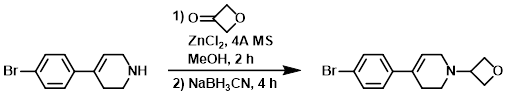
To a solution of the SM (1.10 g, 4.61 mmol) in MeOH (15 mL) was successively added oxetan-3-one (1.66 g, 23.01 mmol), 4A molecular sieves (0.5 g), and ZnCl2 (3.14 g, 23.01 mmol). After stirring 2 h, the reaction mixture was treated with NaBH3CN at 0 C, and stirring was continued for another 4 h. The mixture was diluted with H2O (20 mL), EtOAc (25 mL), and sat aq NaHCO3 (10 mL). The layers were separated and the aq layer was further extracted with EtOAc (3 x 25 mL). The combined organics were washed with brine (50 mL), dried (Na2SO4), and concentrated. The residue was purified by silica gel column chromatography (50% EtOAc/hexane) to provide the product as a colorless syrup. [1.1 g, 81%]
[Patent Reference: WO2015088045, page 108, ![]() (10.3 MB)]
(10.3 MB)]
