Acetyl Deprotection
(Acidic Conditions)
Examples:
Example 1
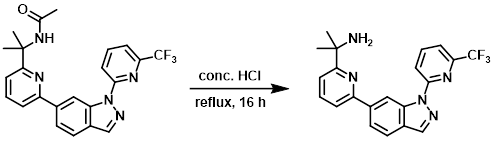
A mixture of the SM (100 mg, 0.23 mmol) in conc. HCl (5 mL) was refluxed for 16 h. After concentration, the residue was basified with sat aq NaHCO3 and extracted with DCM (3 x 30 mL). The combined organics were washed with brine, dried (Na2SO4), and concentrated. The resulting material was purified by Prep HPLC (5-95% MeOH/H2O with 0.05% NH4OH) to provide the product as a brown solid. [70 mg, 77%]
[Patent Reference: WO2016011390, page 210, ![]() (20.2 MB)]
(20.2 MB)]
Example 2
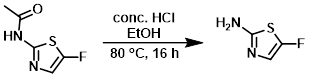
To a solution of the SM (70.0 g, 437 mmol) in EtOH (500 mL) was added conc. HCl (300 mL). The reaction mixture was stirred at 80 C for 16 h. The mixture was concentrated in vacuo and dissolved in H2O. The mixture was neutralized with sat aq NaHCO3 (500 mL) and extracted with EtOAc (2 x 1.0 L). The combined organics were dried (Na2SO4) and concentrated to provide the product which was used for the next reaction without further purification. [15.0 g, crude]
[Patent Reference: WO2014201173, page 72, ![]() (19.7 MB)]
(19.7 MB)]
