SEM Deprotection
(TFA)
Examples:
Example 1

To a solution of the SM (20 mg, 0.03 mmol) in DCM (0.8 mL) was added TFA (0.8 mL). The reaction mixture was stirred at RT for 2 h. The mixture was diluted with DCM (5 mL) and H2O (2 mL). The org layer was dried (Na2SO4) and concentrated. The resulting material was purified by Prep HPLC to provide the product as an oil. [3 mg, 23%]
[Patent Reference: WO2016023832, page 75, ![]() (3.2 MB)]
(3.2 MB)]
Example 2
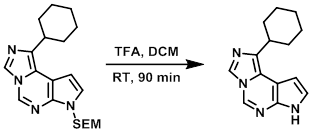
To a solution of the SM (0.16 g, 0.43 mmol) in DCM (3.0 mL) was added TFA (1.0 mL, 13 mmol). After 90 min at RT, the mixture was concentrated in vacuo. The resulting residue was dissolved in dioxane (3.0 mL) and treated with 37% aq NH4OH (3.0 mL, 28.5 mmol). The reaction mixture was stirred at 60 C for 30 min, then concentrated in vacuo. The resulting material was purified by silica gel chromatography [0-100% (970:27:3 DCM/MeOH/diethylamine)/(DCM)] to give the product with ~50 mol% MeOH as an excipient. The solid was dissolved in a minimum amount of hot MeOH (~2 mL) and cooled to RT with sonication. The resulting suspension was concentrated in vacuo and dried in a vac oven at 80 C to provide the product as a tan solid. [50 mg, 48%]
[Patent Reference: WO2012149280, page 54, ![]() (4.1 MB)]
(4.1 MB)]
