Friedel-Crafts
(Acylation)
Examples:
Example 1
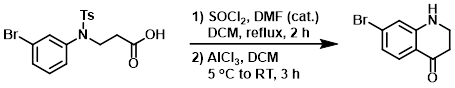
A solution of the SM (5.00 g, 12.59 mmol), SOCl2 (4.60 mL, 62.97 mmol), and DMF (1 drop) in DCM (20 mL) was stirred at reflux for 2 h. The mixture was concentrated to dryness to give the acid chloride. A mixture of AlCl3 (3.40 g, 25.18 mmol) in DCM (20 mL) was cooled to 5 C, then the acid chloride (prepared above) in DCM (10 mL) was added dropwise over 30 min. After addition, the solution was stirred at RT for 3 h. The mixture was quenched with ice H2O, neutralized with NaOH solution, and extracted with EtOAc (3 x 50 mL). The combined organics were dried (Na2SO4) and concentrated. The residue was purified by silica gel column chromatography (1:4 PE/EtOAc) to provide the product as a yellow solid. [1.86 g, 54%]
[Patent Reference: WO2015089337, page 207, ![]() (17.5 MB)]
(17.5 MB)]
