Acetyl Deprotection
(Basic Conditions)
Examples:
Example 1
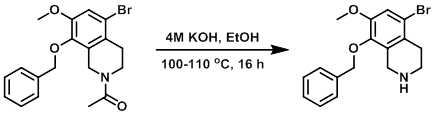
To a solution of the SM (23 g, 59 mmol) in EtOH (100 mL) was added aq 4M KOH (90 mL), and the reaction was stirred at 100-110 C for 16 h. The mixture was concentrated to remove EtOH, and the aq residue was extracted with DCM (2 x 300 mL). The combined org layers were washed with sat aq brine (300 mL), dried (Na2SO4), and concentrated to provide the product as a yellow solid. [17.3 g, 84%]
[Patent Reference: WO2014177977, page 65, ![]() (6.0 MB)]
(6.0 MB)]
Example 2
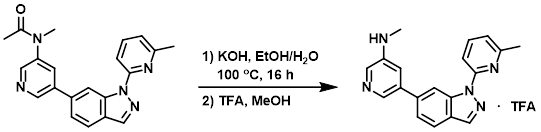
To a solution of the SM (65 mg, 0.18 mmol) in 1:2 EtOH/H2O (3 mL) was added KOH (100 mg, 1.8 mmol). The reaction mixture was heated at 100 C for 16 h. After cooling to RT, the mixture was extracted with DCM (3 x 10 mL). The combined organics were washed with brine (10 mL), dried (Na2SO4), and concentrated. The resulting residue was suspended in MeOH, acidified with TFA, and purified by Prep HPLC to provide the product as a white solid. [30 mg, 62%]
[Patent Reference: WO2016011390, page 64, ![]() (20.2 MB)]
(20.2 MB)]
