SEM Protection
(SEM-Cl + Weak Base)
Examples:
Example 1
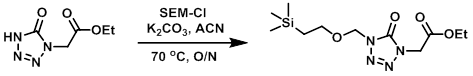
The SM (2.63 g, 15.28 mmol) was dissolved in ACN (75 mL) in a 200 mL, 1-necked, pear-shaped flask equipped with a magnetic stirrer, reflux condenser and Ar inlet. K2CO3 (4.22 g, 30.6 mmol) and SEM-Cl (3.52 mL, 19.86 mmol) were added and the reaction mixture was stirred at 70 C overnight. The mixture was filtered and the filter cake was washed with EtOAc. The filtrate was concentrated in vacuo. The resulting residue was dissolved in a minimal amount of DCM and purified by flash chromagraphy (0-100% EtOAc/hexanes) to provide the product as an oil. [3.61 g, 78%]
[Patent Reference: WO2015191681, page 57, ![]() (6.1 MB)]
(6.1 MB)]
Example 2

To a mixture of the SM (38.7 mg, 0.1 mmol) and K2CO3 (13.8 mg, 0.1 mmol) in DMF (1 mL) at 0 C was added SEM-Cl (33.4 mg, 0.2 mmol). The reaction mixture was stirred at 0 C for 1 h, after which time it was diluted with EtOAc (10 mL) and H2O (3 mL). The layers were separated and the org layer was washed with brine (3 mL), dried (Na2SO4), and concentrated. The resulting material was purified by chromatography (eluting with 5:1 petroleum ether/EtOAc) to provide the product as a colorless oil. [31 mg, 61%]
[Patent Reference: WO2016023832, page 74, ![]() (3.2 MB)]
(3.2 MB)]
Example 3
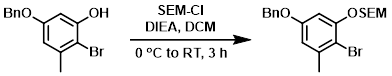
To a solution of the SM (108 g, 0.37 mol) and DIEA (143 g, 1.10 mol) in DCM (1.0 L) at 0 C was added dropwise SEM-Cl (74 g, 0.44 mol). The reaction mixture was stirred at RT for 3 h, then diluted with H2O and extracted with DCM. The org layer was dried and concentrated. The crude product was purified by silica gel chromatography to provide the product. [101 g]
[Patent Reference: WO2016014463, page 133, ![]() (6.7 MB)]
(6.7 MB)]
