CF3 Acetyl Deprotection
(K2CO3)
Examples:
Example 1
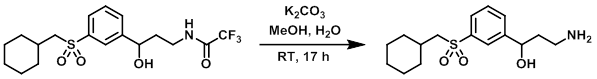
A mixture of the SM (0.472 g, 1.16 mmol), K2CO3 (0.583 g, 4.22 mmol), and 2:1 MeOH/H2O was stirred at RT for 17 h. The mixture was concentrated in vacuo and purified by flash chromatography [30-80% (10% 7N NH3 in MeOH/DCM)/(DCM)] to provide the product as a colorless oil. [0.254 g, 71%] [UK Pat App GB2463151A, page 124]
Example 2
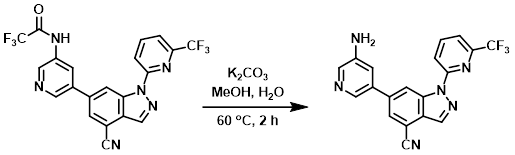
A mixture of the SM (40 mg, 0.09 mmol) and K2CO3 (25 mg, 0.18 mmol) in 2:1 MeOH/H2O (6 mL) was stirred at 60 C for 2 h. After concentration, the residue was diluted with DCM (10 mL) and H2O (5 mL). The org layer was concentrated and the resulting product (yellow solid) was used without further purification. [26 mg, 75%]
[Patent Reference: WO2016011390, page 102, ![]() (20.2 MB)]
(20.2 MB)]
Example 3

To a solution of the SM (60 mg, 0.28 mmol) in MeOH (5 mL) was added K2CO3 (116 mg, 0.84 mmol) at RT. The reaction mixture was stirred at RT for 2 h. After filtration, the mixture was concentrated and purified by Prep HPLC (5-95% MeOH/H2O with 0.05% TFA) to provide the product as a yellow solid. [40 mg, 83%]
[Patent Reference: WO2016011390, page 135, ![]() (20.2 MB)]
(20.2 MB)]
