Aldol Condensation
(Base Catalyzed)
Examples:
Example 1
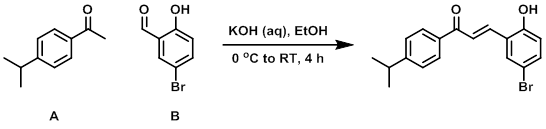
KOH (60% in H2O, 30 mL, 321 mmol) was added to a stirred solution of A (10.00 g, 61.6 mmol) and B (13.6 g, 67.8 mmol) in EtOH (30 mL) at 0 C. The reaction mixture was stirred at RT for 4 h, after which time it was diluted with DCM and quenched with 1N HCl. The resulting precipitate was filtered and washed with H2O and DCM. The filter cake was dried in vacuo to provide the product as a red solid. [19.8 g, 93%]
[Patent Reference: WO2015144799, page 303, ![]() (18.8 MB)]
(18.8 MB)]
Example 2
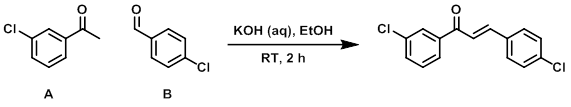
To a solution of A (1.54 g, 10 mmol), B (1.4 g, 10 mmol), and EtOH (25 mL) was added aq KOH (50% wt, 0.56 mL, 10 mmol). The reaction was stirred at RT for 2 h. The resulting solids were filtered and washed with 5:1 PE/EtOAc (10 mL) to provide the product as a white solid which was used without further purification. [2.3 g, 83%]
[Patent Reference: WO2016023832, page 71, ![]() (3.2 MB)]
(3.2 MB)]
Example 3
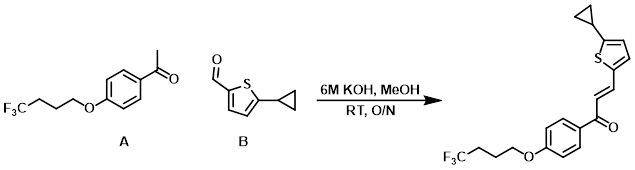
To a solution of the A (250 mg, 1.015 mmol) and B (155 mg, 1.015 mmol) in MeOH (20 mL) at RT was added KOH (1.33 g, 23.71 mmol) and H2O (3 mL). The reaction mixture was stirred at RT overnight. The mixture was filtered and dried under suction to provide the product as a solid. [225 mg, 58%]
[Patent Reference: WO2015191681, page 59, ![]() (6.1 MB)]
(6.1 MB)]
