Alcohol to Mesylate
(MeSO2Cl)
Examples:
Example 1
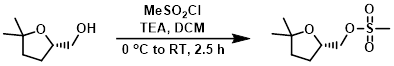
To a solution of the SM (300 mg, 2.30 mmol) in DCM (6 mL) at 0 C was added TEA (0.64 mL, 4.6 mmol) followed by MeSO2Cl (0.21 mL, 2.76 mmol). The reaction mixture was stirred at 0 C for 30 min, then was allowed to warm to RT over 2 h. The mixture was quenched with H2O (10 mL) and extracted with DCM (2 x 20 mL). The combined organics were washed with H2O (20 mL), brine (20 mL), dried (Na2SO4), and concentrated. The resulting material was purified by silica gel column chromatography (35% EtOAc/hexane) to provide the product as a colorless gum. [310 mg, 64%]
[Patent Reference: WO2015129926, page 89, ![]() (21.5 MB)]
(21.5 MB)]
Example 2
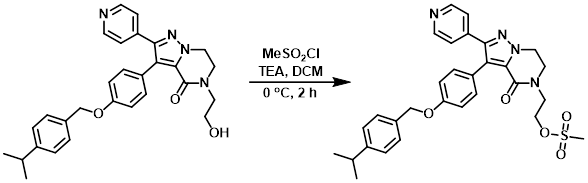
MeSO2Cl (49 uL, 0.64 mmol) was added dropwise to a solution of the SM (205 mg, 0.43 mmol) and TEA (178 uL, 1.3 mmol) in dry DCM (5 mL) at 0 C under N2. The reaction mixture was stirred at 0 C for 2 h, after which time H2O was added and the mixture was extracted with DCM. The org layer was dried (MgSO4) and concentrated to provide the product as a yellow solid which was taken to the next step without further purification. [280 mg]
[Patent Reference: WO2015144799, page 153, ![]() (18.8 MB)]
(18.8 MB)]
Example 3

To a solution of the SM (2.50 g, 16.4 mmol) in DCM (20 mL) was added TEA (2.75 mL, 19.7 mmol) followed by MeSO2Cl (1.53 mL, 19.7 mmol). The reaction mixture was stirred at RT overnight. The mixture was diluted with H2O and extracted with DCM. The combined organics were washed with brine, dried (MgSO4), and concentrated to provide the product which was taken to the next step without further purification.
[Patent Reference: WO2016014463, page 122, ![]() (6.7 MB)]
(6.7 MB)]
Example 4

To a solution of the SM (0.5 g, 2.17 mmol) in DCM (13 mL) was added TEA (0.6 mL, 4.34 mmol) and MeSO2Cl (0.2 mL, 2.6 mmol) in DCM (2 mL) at 0 C. The reaction mixture was stirred at RT for 1 h. The mixture was diluted with DCM and H2O. The org layer was separated, dried (Na2SO4), and concentrated. The residue was purified by silica gel column chromatography to provide the product as a syrup. [0.3 g]
[Patent Reference: WO2014149164, page 398, ![]() (23.7 MB)]
(23.7 MB)]
