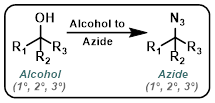
Alcohol to Azide
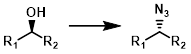
Common Conditions:
Mitsunobu
Mitsunobu conditions convert alcohols to azides with inversion of stereochemistry. The separation of phosphine oxide impurities can be difficult.[1]
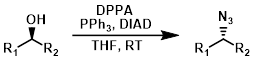
DPPA + DBU
Only reactive alcohols such as benzylic alcohols react well under these conditions. A major benefit is that no difficult to remove phosphine oxides (ex. PPh3O) form in this reaction. These conditions also provide SN2 inversion of configuration.[1][2]
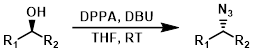
Reaction Map:
The reaction map is intended to provide insight into possible reactions one step before and after the title reaction. It also serves as an alternative way to navigate the website, and as a means of coming up with retrosynthetic ideas. Click on the reaction arrow to visit the page.
 |
||||
 |
||||
 |
 |
 |
||
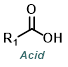 |
||||
 |
||||
 |
References:
1) TCI Chemicals, Diphenyl Phosphorazide (DPPA) - More Than Three Decades Later (link)
2) Thompson, A. S.; WO1995001970, Alcohol to Azide SN2 Conversion (link)
