Alcohol to Azide
(DPPA + DBU)
Examples:
Example 1
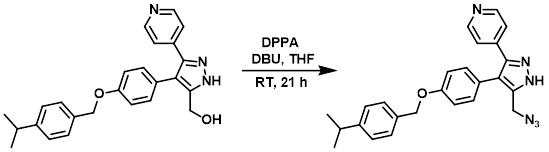
To a suspension of the SM (0.665 g, 1.66 mmol) in THF (10 mL) at RT under Ar was added DPPA (0.400 mL, 1.83 mmol) and DBU (0.297 mL, 1.99 mmol). The resulting mixture was stirred at RT for 18 h. Additional DPPA (0.200 mL, 0.916 mmol) and DBU (0.150 mL, 1.00 mmol) was added and the mixture was stirred at RT for 3 h. The reaction mixture was quenched with sat aq NaHCO3 (30 mL). The aq mixture was extracted EtOAc (2 x 30 mL). The combined organics were dried (Na2SO4) and concentrated to a yellow gum. The material (1.26 g) was purified by Prep LC (2-5% MeOH/DCM) to provide the product as an off-white solid. [370 mg, 52%]
[Patent Reference: WO2015144799, page 343, ![]() (18.8 MB)]
(18.8 MB)]
Example 2

A solution of the SM (600 mg, 2.5 mmol) in toluene (10 mL) was cooled in an ice bath under N2 and treated with DPPA (2.06 g, 7.5 mmol) in one portion, followed by DBU (1.14 g, 7.5 mmol). The reaction mixture was stirred at 0 C for 1 h, then RT for 12 h. The mixture was diluted with EtOAc (100 mL), washed with 2N HCl (2 x 50 mL), brine, dried (Na2SO4), and concentrated. The crude product was purified by silica gel column chromatography (eluted with PE) to provide the product as a yellow oil. [350 mg, 45%]
[Patent Reference: WO2015089337, page 184, ![]() (17.5 MB)]
(17.5 MB)]
