Acid to Ester
(MeI)
Examples:
Example 1
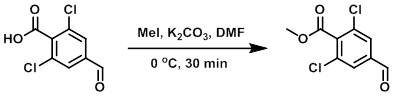
To a solution of the SM (1.1 g, 5.0 mmol) in DMF (10 mL) at 0 C was added K2CO3 (1.0 g, 7.5 mmol) followed by the slow addition of MeI (0.94 mL, 15.0 mmol), and the reaction was stirred at the same temperature for 30 min. The reaction mixture was quenched with H2O (50 mL) and extracted with EtOAc (2 x 30 mL). The combined organics were washed with H2O (50 mL), brine (50 mL), dried (Na2SO4), and concentrated. The resulting material was purified by silica gel column chromatography (10% EtOAc/hexane) to provide the product as a white solid. [0.59 g, 50%]
[Patent Reference: WO2015129926, page 95, ![]() (21.5 MB)]
(21.5 MB)]
Example 2
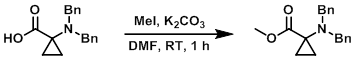
To a solution of the SM (21.0 g, 74.7 mmol) in DMF (63.0 mL) at 0 C was added K2CO3 (31.0 g, 224.0 mmol) and MeI (11.6 mL, 187.0 mmol). The reaction mixture was stirred at RT for 1 h. Upon completion, the reaction mixture was quenched with H2O, extracted with EtOAc, dried (Na2SO4), and concentrated in vacuo to provide the product as a colorless liquid. [22.0 g, 99%]
[Patent Reference: WO2014149164, page 233, ![]() (23.7 MB)]
(23.7 MB)]
Example 3
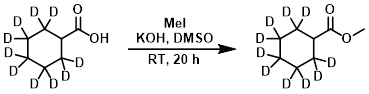
To a solution of the SM (1.71 g, 12.3 mmol) in dry DMSO was added finely powdered KOH (729 mg, 13.0 mmol). The mixture was stirred at RT for 25 min, after which time was added MeI (1.96 g, 13.8 mmol). The reaction mixture was stirred at RT for 20 h, then partitioned between H2O and ether. The org layer was separated, washed with brine, treated with activated charcoal, dried (MgSO4), and concentrated in vacuo to provide the product as a colorless oil. [1.86 g, 99%] [UK Pat App GB2463151A, page 157]
