Amide to Amine
(LiAlH4)
Examples:
Example 1
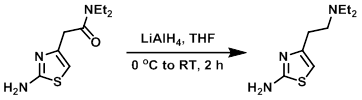
A suspension of the SM (1 eq) in THF was added dropwise to a vigorously stirred suspension of LiAlH4 (1 eq) in THF at 0 C. The reaction mixture was stirred at RT for 2 h. Upon completion, the mixture was cooled to 0 C and treated sequentially with 1 part H2O, 1 part 10% NaOH, and 3 parts H2O in a dropwise manner. The mixture was stirred for 10 min, filtered, and the filter cake washed with THF. The filtrate was concentrated and the resulting crude washed with Et2O (2x) and dried to provide the product as a viscous brown oil.
[Patent Reference: WO2007084786, page 143, ![]() (9.4 MB)]
(9.4 MB)]
Example 2
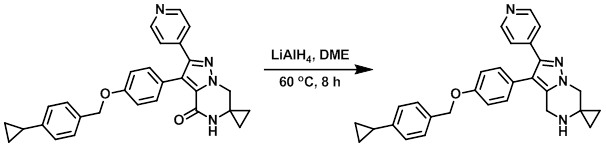
LiAlH4 (98.5 mg, 2.59 mmol) was added to a solution of the SM (200 mg, 0.432 mmol) in dry DME (8 mL). The reaction mixture was stirred at 60 C for 8 h, after which time it was very slowly quenched with H2O (100 uL) and 3N NaOH (100 uL). Ether was added and the mixture was filtered through a glass frit. The solids were washed with ether and the filtrate was concentrated in vacuo to give 330 mg of a yellow material. The material was purified by Prep LC (24 g silica, 0-50% acetone/DCM) to provide the product as a white solid. [118 mg, 49%]
[Patent Reference: WO2015144799, page 278, ![]() (18.8 MB)]
(18.8 MB)]
