Alkene to Alkane
(H2 + PtO2)
Examples:
Example 1
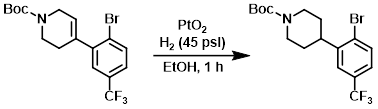
A solution of the SM (2.435 g, 5.99 mmol) in EtOH (20 mL) was treated with PtO2 (0.136 g, 0.599 mmol), and was placed under 45 psi H2 for 1 h. LCMS indicated mostly product and de-brominated product. The mixture was diluted with DCM and filtered through celite. The filtrate was concentrated then purified directly by column chromatography (0-15% EtOAc/heptane) to provide the product. [0.970 g, 39.6%]
[Patent Reference: WO2014201173, page 63, ![]() (19.7 MB)]
(19.7 MB)]
Example 2
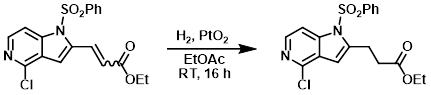
To a mixture of the SM (0.7 g, 1.79 mmol) in EtOAc (25 mL) was added PtO2 (0.150 g) at RT. The reaction mixture was stirred under H2 atmosphere (balloon pressure) at RT for 16 h. The catalyst was removed by filtration through a celite pad and washed with EtOAc. The filtrate was concentrated and purified by silica gel column chromatography (20% EtOAc/hexane) to provide the product as a light yellow solid. [0.610 g, 86%]
[Patent Reference: WO2015088045, page 163, ![]() (10.3 MB)]
(10.3 MB)]
Example 3
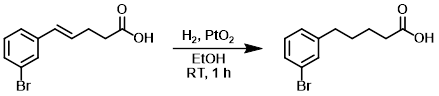
To a solution of the SM (2.4 g, 9.4 mmol) in EtOH (20 mL) was added PtO2 (10%, 200 mg). The mixture was stirred for 1 h under a H2 atmosphere. The catalyst was filtered out and the filtrate was concentrated to provide the product as a yellow solid which was used in the next step without further purification. [2.1 g, 87%]
[Patent Reference: WO2015089337, page 183, ![]() (17.5 MB)]
(17.5 MB)]
