Acid to Alcohol
(LiAlH4)
Examples:
Example 1
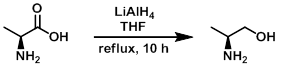
LiAlH4 (12.78 g, 336.7 mmol) was suspended in dry THF (550 mL) at 0 C. To this mixture was added the SM (15.0 g, 168.4 mmol) in portions over 20 min. The mixture was then refluxed for 10 h. After cooling the mixture to 0 C it was diluted with 2N NaOH (70 mL) and stirred at RT for 3 h. The resulting solids were filtered and washed with THF (200 mL). The solids were then suspended in THF (250 mL) and refluxed for 1 h. The solids were filtered and washed with THF (100 mL). The same trituration procedure was repeated twice and the resulting THF solutions (containing the product) were combined and used immediately in the next reaction.
[Patent Reference: WO2003061598, page 23, ![]() (3.5 MB)]
(3.5 MB)]
Example 2

A solution of the SM (350 g, 2.10 mol) in THF (1.4 L) was added to a slurry of LiAlH4 (95.9 g, 1.40 mol) in THF (2.5 L) at 0 C. The reaction mixture was stirred at RT for 30 min, then heated to reflux for 60 min. The mixture was cooled to 0 C and slowly quenched by the addition of sat aq NH4Cl. A large excess of solid Na2SO4 and EtOAc were added. The solids were removed by filtration and the filtrate was concentrated in vacuo to provide the product which was taken to the next step without further purification. [350 g, quant.]
[Patent Reference: WO2016014463, page 90, ![]() (6.7 MB)]
(6.7 MB)]
