Phosphorus Oxychloride
Other Names:
Phosphoryl chloride
Phosphoryl trichloride
Phosphorus(V) oxide chloride
General Information:
Structure:
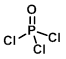
CAS Number: 10025-87-3
Molecular Weight: 153.33 g/mol
Appearance: Colorless liquid
Chemical Formula: POCl3
Melting Point: 1.25 C
Boiling Point: 105.8 C
Density: 1.645 g/mL
Common Uses:
Reagent for the formation of active chloro-heterocycles
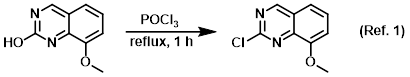
Procedure excerpt:
Neat POCl3 (400 mL) was added to the SM (5.0 g, 28.4 mmol) with stirring at 0 C under Ar. After 1 min, the reaction was removed from the ice bath and stirred at RT . . .
Reagent for the dehydration of unsubstituted amides to nitriles
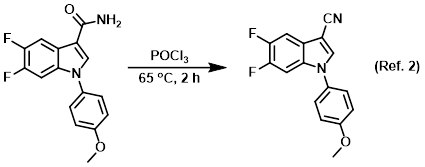
Procedure excerpt:
A solution of the SM (420 mg, 1.39 mmol) in POCl3 (15 mL) was stirred at 65 C for 2 h. The mixture was cooled to RT and diluted with toluene. The volatiles were . . .
Reagent in Bischler-Napieralski reactions
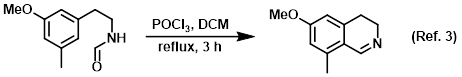
Procedure excerpt:
To a solution of the SM (76.0 g, 0.354 mol) in DCM (2.5 L) at 15 C was added POCl3 (155 g, 1.01 mol). After addition, the reaction mixture was heated to reflux . . .
Safety:
Phosphorus oxychloride (POCl3) is very toxic and reacts strongly with water.
References:
1) Patent Reference: WO2007117607, page 307, ![]() (12.9 MB)
(12.9 MB)
2) Patent Reference: WO2010031852, page 37, ![]() (2.8 MB)
(2.8 MB)
3) Patent Reference: WO2016014463, page 94, ![]() (6.7 MB)
(6.7 MB)
4) Wikipedia: Phosphoryl chloride (link)
5) www.sigmaaldrich.com: Phosphorus(V) oxychloride (link)
6) Pearson, A. J.; Roush, W. R.; Handbook of Reagents for Organic Synthesis; Activating Agents and Protecting Groups
