Activated Cl Formation
(POCl3)
Examples:
Example 1
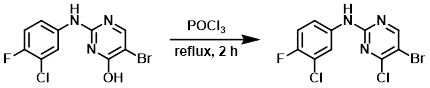
A solution of the SM (6.5 g, 18.3 mmol) in POCl3 (21 mL) was heated to reflux for 2 h, after which time it was cooled to RT and poured carefully into a stirring mixture of ice (200 mL) and saturated aq NaHCO3 (20 mL). The resulting mixture was extracted with EtOAc (2 x 250 mL). The combined org layers were washed with H2O, brine, dried (Na2SO4), and concentrated in vacuo. The residue was purified by silica gel column chromatography (3% EtOAc/hexane) to provide the product. [5.1 g, 15 mmol]
[Patent Reference: WO2010038081, page 88, ![]() (33.8 MB)]
(33.8 MB)]
Example 2
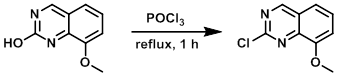
Neat POCl3 (400 mL) was added to the SM (5.0 g, 28.4 mmol) with stirring at 0 C under Ar. After 1 min, the reaction was removed from the ice bath and stirred at RT for 20 min until a fine yellow suspension formed. A reflux condenser was added and the mixture was stirred at 140-145 C for 1 h, after which time the reaction became a colorless and clear solution. LCMS indicated a complete reaction. The mixture was concentrated in vacuo to remove POCl3 and the resulting crude was dried in vacuo. After 12 h, the crude was then partitioned between EtOAc (300 mL) and sat aq NaHCO3 (200 mL) and stirred cautiously (watching gas evolution) until pH 8. The org layer was separated, washed with NaHCO3 (2 x 100 mL), H2O with 5% brine (2 x 100 mL), brine (100 mL), dried (Na2SO4), concentrated, and purified by flash chromatography (50-100% EtOAc/hexane) to provide the product as a white solid. [4.9 g, 89%]
[Patent Reference: WO2007117607, page 307, ![]() (12.9 MB)]
(12.9 MB)]
Example 3
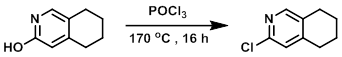
A suspension of the SM (2.0 g, 13 mmol) and POCl3 (10.0 mL) was stirred in a sealed tube at 170 C for 16 h. Upon completion, the reaction mixture was poured into ice/H2O, neutralized with sat aq NaHCO3, and extracted with EtOAc. The org layer was dried (Na2SO4) and concentrated in vacuo. The resulting material was purified by silica gel column chromatography (5% EtOAc/hexane) to provide the product as a light yellow liquid. [850 mg, 38%]
[Patent Reference: WO2014149164, page 240, ![]() (23.7 MB)]
(23.7 MB)]
Example 4
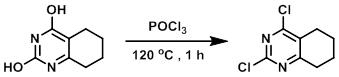
A mixture of the SM (8.0 g) and POCl3 (40 mL) was stirred at 120 C for 1 h. Upon completion, the reaction mixture was cooled to RT and the excess POCl3 was removed in vacuo. The resulting material was partitioned between EtOAc (400 mL) and H2O (200 mL). The org layer was washed with sat aq NaHCO3 (200 mL), brine (200 mL), dried (Na2SO4), and concentrated in vacuo. The resulting material was purified by column chromatography to provide the product as a yellow solid. [4.0 g, 57%]
[Patent Reference: WO2014149164, page 250, ![]() (23.7 MB)]
(23.7 MB)]
