Nitro Reduction
(Na2S)
Examples:
Example 1
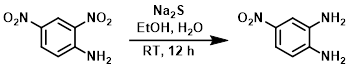
To a solution of the SM (1 g, 10.3 mmol) in 3:1 EtOH/H2O (26 mL) was added Na2S (1.2 g, 15.4 mmol). The reaction mixture was stirred at RT for 12 h. After completion of the reaction, solvent was removed in vacuo. The resulting residue was diluted with H2O and extracted with EtOAc. The org layer was dried (Na2SO4) and concentrated. The resulting material was purified by silica gel column chromatography to provide the product as a brown solid. [200 mg, 24%]
[Patent Reference: WO2014149164, page 480, ![]() (23.7 MB)]
(23.7 MB)]
Example 2
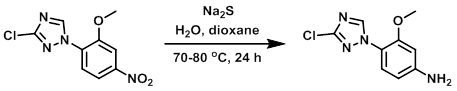
H2O (8 mL) and dioxane (8 mL) were added to a mixture of the SM (900 mg, 3.53 mmol) and Na2S (1.379 g, 17.67 mmol) in a 20 mL vial. The vial was capped and heated at 70-80 C for 24 h. The mixture was cooled to RT, poured into H2O (300 mL) and extracted with EtOAc (2 x 150 mL). The combined organics were washed with brine, dried (Na2SO4), and concentrated to provide the product as a brown solid. [577 mg, 73%]
[Patent Reference: WO2011014535, page 47, ![]() (17.3 MB)]
(17.3 MB)]
