Formylation
(Rieche Formylation)
Examples:
Example 1
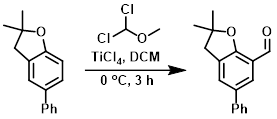
To a 0 C solution of the SM (2.24 g, 10.0 mmol) in DCM (15 mL) was added slowly TiCl4 (2 mL, 18 mmol). After stirring 5 min, dichloro(methoxy) methane (1 mL, 11 mmol) was added slowly. The resulting mixture was stirred at 0 C for 3 h, after which time it was quenched slowly with ice H2O. The mixture was extracted with DCM. The org layer was separated, dried (Na2SO4), concentrated, and purified by silica gel flash chromatography (5-10% EtOAc/hexane) to provide the product.
[Patent Reference: WO2011159297, page 101, ![]() (10.6 MB)]
(10.6 MB)]
Example 2
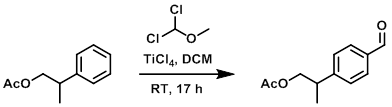
A solution of the SM (3.0 g, 16.8 mmol) and dichloro(methoxy) methane (3.87 g, 33.7 mmol) in dry DCM (15 mL) was cooled to 0 C and treated with TiCl4 (1M in DCM, 84 mL, 84 mmol) over 15 min. The reaction mixture was then warmed to RT and stirred for 17 h. The mixture was poured into ice and DCM was added. The org layer was separated, washed with brine, dried (MgSO4), and concentrated to a black oil which was taken to the next step without further purification. [3.8 g, quant.]
[Patent Reference: WO2015144799, page 229, ![]() (18.8 MB)]
(18.8 MB)]
