Boc Protection
(Boc2O + DMAP)
Mechanism:
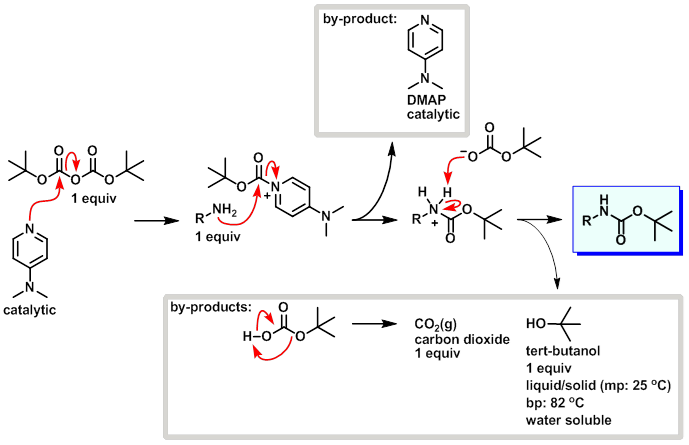
Steps:
- DMAP attacks a carbonyl site on di-tert-butyl dicarbonate (Boc2O) resulting in tert-butyl carbonate leaving as a leaving group.
- The amine attacks the carbonyl site on the boc-pyridinium species resulting in the release of DMAP (catalytic).
- tert-Butyl carbonate picks up the proton from the protonated amine.
- tert-Butyl bicarbonate breaks down into CO2 (gas) and tert-butanol.
Key Points:
- The reaction of DMAP with Boc2O is almost instantaneous (gas formation!). To avoid rapid gas formation slowly add the DMAP over an appropriate period of time.
- The CO2 gas that forms during the reaction should be allowed to escape. Don't run boc protections in closed systems.
- DMAP should accelate the rate of the boc protection reaction but also makes side reactions more likely.
