Alcohol to Acid
[TEMPO (cat.) + NaOCl (cat.)
+ NaClO2]
Examples:
Example 1
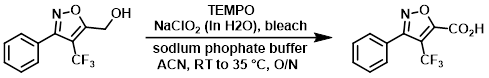
A fresh solution of sodium phosphate buffer (40 mL, pH ~6.5) consisting of a 1:1 solution of NaH2PO4 (20 mL, 0.67 M) and Na2HPO4 (20 mL, 0.67 M) was prepared. A mixture of the SM (2.1 g, 8.64 mmol), TEMPO (0.094 g, 0.604 mmol), and sodium phosphate buffer (32.2 mL, 21.59 mmol, 0.67 M) in ACN (30 mL) heated to 35 C. Solutions of NaClO2 (3.91 g, 34.5 mmol) in H2O (4.5 mL) and bleach (4.3 mL, 6% wt) were added simultaneously over 40 min. The reaction was monitored by HPLC. After 2 h, ~30% SM remained. After 6 h, ~10% SM remained. Additional bleach (100 uL) was added, and the reaction was left at RT overnight. Additional bleach (100 uL) was added. The resulting mixture was stirred at 35 C for 2 h, after which time HPLC indicated a complete conversion. The reaction mixture was quenched by the slow addition of solution of Na2SO3 (2.07 mL, 43.2 mmol) in H2O (90 mL) at 0 C, resulting in the disappearance of the brown reaction color. The solvent was removed in vacuo, and the remaining aq residue was extracted with EtOAc (3 x 40 mL). The organics were combined, washed with H2O (8 mL), brine (8 mL), dried (Na2SO4), and concentrated to provide the product as a pale yellow solid. [2.2 g, 99%]
[Patent Reference: WO2011017578, page 71, ![]() (8.3 MB)]
(8.3 MB)]
