Acid to Ester
(Me2SO4)
Examples:
Example 1
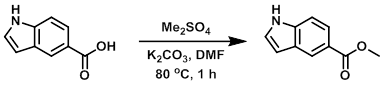
A solution of the SM (1.0 g, 6.2 mmol), K2CO3 (0.939 g, 6.8 mmol), and DMF (10 mL) was heated to 65 C and treated dropwise with Me2SO4 (0.78 g, 6.2 mmol) over 15 min, keeping the temperature between 65-70 C. The reaction was stirred at 80 C for 1 h, after which time it was poured into ice/water. The resulting solids were collected by filtration and dried in vacuo to provide the product. [0.91 g, 86%]
[Patent Reference: WO2012129338, page 67, ![]() (12.0 MB)]
(12.0 MB)]
Example 2
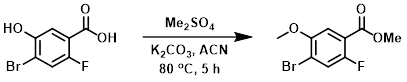
To a solution of the SM (3.5 g, 14.9 mmol) in ACN (40 mL) was added K2CO3 (6.16 g, 44.7 mmol) and Me2SO4 (5.63 g, 44.7 mmol) at RT. The reaction mixture was stirred at 80 C for 5 h, after which time TLC (2:8 EtOAc/PE) indicated a complete consumption of starting material. The mixture was cooled to RT, diluted with H2O (250 mL), and extracted with EtOAc (3 x 100 mL). The combined organics were washed with brine (100 mL), dried (Na2SO4), and concentrated. The crude material was purified by silica gel column chromatography (5% EtOAc/PE) to give impure product. The impure material was triturated with ether (20 mL) to provide the product as an off white solid. [1.5 g, 38%]
[Patent Reference: WO2015051043, page 63, ![]() (9.7 MB)]
(9.7 MB)]
