Boron Trichloride
Other Names:
Boron(III) chloride
Trichloroborane
General Information:
Structure:
![]()
CAS Number: 10294-34-5
Molecular Weight: 117.17 g/mol
Appearance: Colorless gas
Chemical Formula: BCl3
Melting Point: -107 C
Boiling Point: 12.5 C
Boron trichloride (BCl3) is a Lewis acid typically used for the demethylation of methyl ethers. Boron trichloride is not as reactive as boron tribromide (BBr3), but this lower reactivity can sometimes be beneficial for controlling selectivity. At room temperature boron trichloride is gas, but usually it is purchased and used as a 1M solution in DCM.
Common Uses:
Reagent for the demethylation of methyl ethers
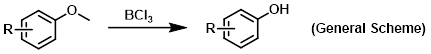
Safety:
Boron trichloride (BCl3) reacts violently and exothermically with water and moisture in the air. Always handle under inert atmosphere.
References:
1) Wikipedia: Boron trichloride (link)
2) www.sigmaaldrich.com: Boron trichloride solution (link)
3) Reich, H. J.; Rigby, J. H.; Handbook of Reagents for Organic Synthesis, Acidic and Basic Reagents
