SNAr (F)
(N-Heteroaryls)
Examples:
Example 1
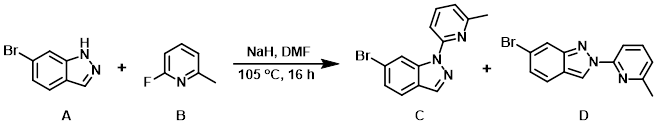
To a solution of the indazole (A) (3.0 g, 15.3 mmol) in DMF (10 mL) at 0 C was added NaH (440 mg, 18.36 mmol). The mixture was stirred at RT for 1 h. The aryl fluoride (B) (2.04 g, 18.36 mmol) was added and the reaction mixture was stirred at 105 C for 16 h. After cooling to RT, the mixture was diluted with H2O (50 mL) and stirred 1 h. The resulting mixture was filtered to give a yellow solid which was purified by prep TLC (10:1 PE/EtOAc) to provide product C (1.0 g, 23%) as a yellow solid and product D (1.05 g, 24%) as a yellow solid. [total yield: 2.05 g, 47%]
[Patent Reference: WO2016011390, page 54, ![]() (20.2 MB)]
(20.2 MB)]
