DCE
(1,2-Dichloroethane)
Other Names:
Ethylene dichloride
Ethane dichloride
General Information:
Structure:
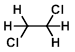
CAS Number: 107-06-2
Molecular Weight: 98.96 g/mol
Appearance: Colorless liquid
Melting Point: -35 C
Boiling Point: 83 C
Density: 1.256 g/mL at 25 C
1,2-Dichloroethane (DCE) is a generally used as a solvent in organic chemistry. DCE can act as an alkylating agent in the presence of a suitable nucleophile and under the right reaction conditions.
Common Uses:
Solvent for reactions
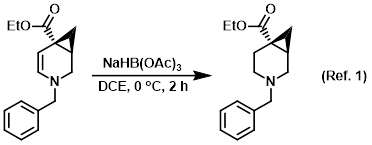
Procedure excerpt:
To a solution of the SM (crude) in DCE (50 mL) at 0 C was added NaBH(OAc)3 (3.6 g, 17.1 mmol). The reaction mixture was stirred at 0 C for 2 h, after which time . . .
Safety:
1,2-Dichloroethane (DCE) is a toxic, carcinogenic, and highly flammable liquid.
References:
1) Patent Reference: WO2016014463, page 64, ![]() (6.7 MB)
(6.7 MB)
2) Wikipedia: 1,2-Dichloroethane (link)
3) www.sigmaaldrich.com: 1,2-Dichloroethane (link)
