THP Deprotection
(p-TsOH)
Examples:
Example 1

A mixture of the SM (2.05 g, 6.8 mmol) and p-TsOH monohydrate (260 mg, 1.35 mmol) in MeOH (60 mL) under N2 was stirred at RT for 16 h. The mixture was diluted with EtOAc and washed with sat aq NaHCO3. The org layer was separated, dried (Na2SO4), and concentrated to provide the product. [1.41 g, 95%]
[Patent Reference: WO2005082859, page 297, ![]() (25.9 MB)]
(25.9 MB)]
Example 2
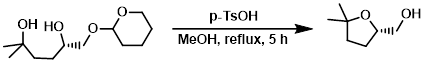
To a solution of the SM (1.03 g, 4.43 mmol) in MeOH (8 mL) at RT was added p-TsOH monohydrate (421 mg, 2.2 mmol). The reaction mixture was stirred at reflux for 5 h, after which time it was cooled to RT. The mixture was quenched with H2O (15 mL) and extracted with DCM (2 x 25 mL). The combined organics were washed with brine (20 mL), dried (Na2SO4), and concentrated. The resulting material was purified by silica gel column chromatography (35% EtOAc/hexane) to provide the product as a colorless gum. [330 mg, 57%]
[Patent Reference: WO2015129926, page 89, ![]() (21.5 MB)]
(21.5 MB)]
